 The Therapeutic Goods Administration (TGA) has provisionally approved the Spikevax (Moderna) vaccine for children aged 6 months and over. Read the latest advice and recommendations for booster doses. The booster dose is given 6 months after the last dose or COVID-19 infection, whichever is more recent. Moderna can also be used as a booster dose in people aged 18 years and older. It is usually given 8 weeks after the second dose of the primary course. Some people who are severely immunocompromised are recommended to have a third dose as part of their primary course. Moderna can be used for this third dose. You may not be protected against COVID-19 until 14 days after a vaccine dose. A shorter interval may be recommended for people at higher risk of severe COVID-19 (including older adults and people with underlying medical conditions), or before international travel. It is preferable to have the first 2 doses 8 weeks apart, but the dose interval between doses can be reduced to 4 weeks. Dose schedule Primary courseįor most people the Moderna primary course is 2 doses, 8 weeks apart. However, both the bivalent and original vaccines provide significant protection from severe disease against Omicron subvariant infections. The Moderna bivalent COVID-19 vaccine targets the original 2020 strain and the Omicron variant BA.1.Ĭompared to the original vaccines, ATAGI found it triggers a modest improvement in the immune response against both strains. The Australian Technical Advisory Group on Immunisation (ATAGI) has provided a recommendation that the Moderna bivalent vaccine can be used as an alternative vaccine for any booster dose in people aged 18 years or older, according to the current ATAGI recommendations for booster doses. On 29 August, the TGA provisionally approved Moderna's bivalent COVID-19 vaccine for use as a booster dose in adults 18 years and over, and this vaccine has been available since 10 October 2022. On 21 April 2023, the TGA approved Moderna Australia’s application to transition its COVID-19 vaccine, SPIKEVAX (elasomeran), from provisional to full registration. 19 July 2022 (primary course – for children 6 months and over) (6 month to 5 year formulation)ģ0 August 2022 (booster – for 18 years and over) (bivalent original/Omicron BA.1 formulation).17 February 2022 (primary course - for 6 years and over) (half the adult dose). 
7 December 2021 (booster – for 18 years and over).3 September 2021 (primary course - for 12 years and over).9 August 2021 (primary course - for 18 years and over).The Therapeutic Good Administration (TGA) provisionally approved it for use in Australia on:  
Moderna is approved and available for use as a booster in people aged 18 years and over. Spikevax (Moderna) is approved and available for use as a primary course in people aged 6 months and over. “But now there’s more choice and we need to evaluate the data and make a halftime adjustment.”įor some, the decision to mix and match the booster may be driven by concerns about very rare but serious side effects tied to each vaccine: The Johnson & Johnson vaccine has been linked to a very slight increased risk of blood clots in women under 50, and the Pfizer and the Moderna vaccines have been linked to an increased risk of a heart condition called myocarditis, particularly for males under 30.Information on this page has been superseded by new ATAGI advice on the preferential use of bivalent COVID-19 vaccines for primary course and will be updated shortly. “When the vaccines were first introduced, the Johnson & Johnson shot did have its benefits and there wasn’t as much supply, so people needed to get whichever vaccine was available,” Sobhanie said. People who chose Johnson & Johnson’s single-dose vaccine out of convenience or people in transient populations who could not commit to a second dose may now be opting for an mRNA booster since the benefits can be delivered in just one booster shot. (The Pfizer and the Moderna vaccines were the first ever authorized using mRNA.) 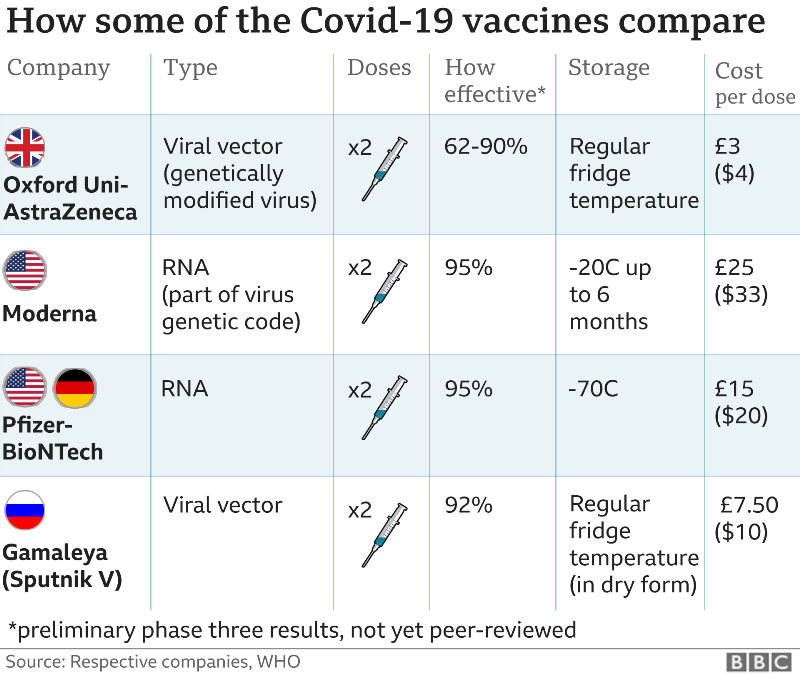
Those who were originally wary of the mRNA technology early on may also be ready to adopt it now, Limaye said. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
